 The bromine that was just added forms two bonds (one on each carbon of the double bond), giving a three-membered C-Br-C ring called a bromonium ion (since the bromine atom now has a positive charge). In this type of reaction (electrophilic addition to an alkene), the ∏-bond (double bond) on ethene attacks a bromine atom (from Br2) and kicks out a bromide (Br-). In the absence of light or heat, bromine cannot react with an alkane, but it can react as an electrophile with an alkene. The termination step leading to the product is one where another bromine radical joins with the ethyl radical. 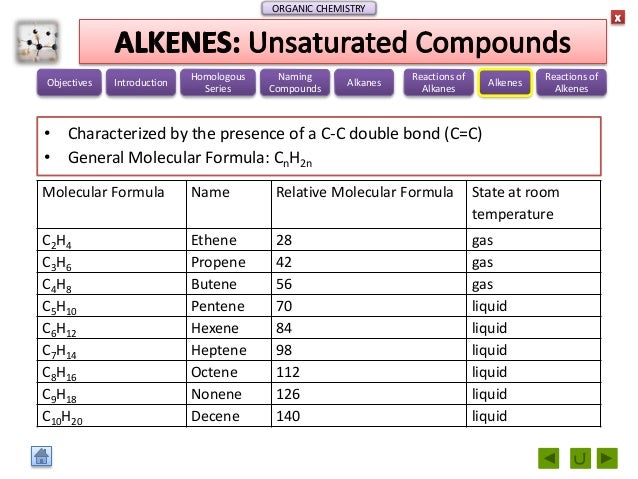
This goes through an initiation step (forming 2 bromine radicals), followed by propagation to the alkane (forming a secondary ethyl radical), followed by a termination step. Br2 in the presence of a radical initiator (such as light or heat) will add to ethane to form 2-bromoethane as the major product in a radical mechanism. The question is not very specific, so there is more than just one answer, but I'm assuming you are referring to a radical bromination of an alkane (ethane) versus an electroph ilic bromination of an alkene (ethene). 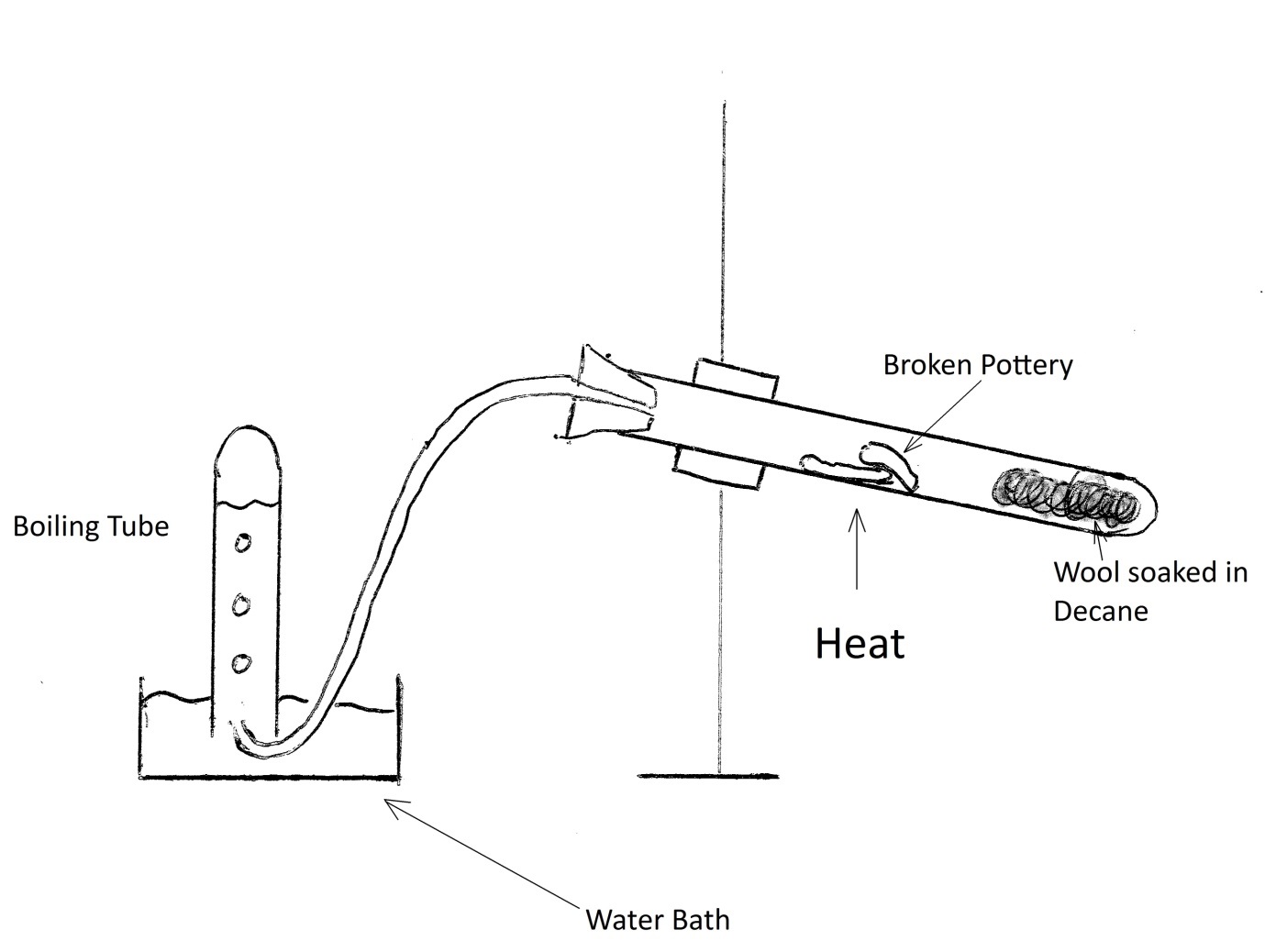
A secondary school revision resource for AQA GCSE Science about the Earth, fats, polymers and ethanol from oils. Catalytic cracking is acheived by the use of a catalyst and lower temperatures. Pentyl radicals -> Propyl radical + Ethene. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
